The best results for Western blots are obtained when both the primary and secondary antibodies are accurately titrated. Although dot blots cannot determine the molecular weight or integrity of a protein and therefore should never be used to identify a protein per se, they are particularly useful in titrating antibodies. Optimal antibody concentrations can be efficiently determined by adhering proteins to nitrocellulose using a dot blot technique with a checkerboard pattern to determine the optimal primary: secondary concentration pair.
Below are guidelines and general protocols for performing dot blots either by using a microfiltration unit or by manually spotting protein onto a membrane.
Dot blot Overview
Dot blots are similar to Western blots, however the proteins are not separated electrophoretically prior to transfer to a membrane but are instead spotted directly onto a membrane. Because proteins are not first separated using a gel, dot blots cannot be used to determine the molecular weight of a protein nor can they discriminate between alternate forms of the protein (e.g. cleaved proteins). However when the integrity and identity of the protein is known, the dot blot format can be used to provide substrates for titration of antibodies.
Commercially available units for dot blotting
Dot blots can be performed using commercially available apparatuses, often called microfiltration units. Microfiltration units provide an ease of use as protein blotting, incubations and washes can all be performed within the unit which isolates each individual blot.
Manual dot blots
Dot blots can also be performed without the aid of a microfiltration unit. In this case, protein is spotted manually onto the membrane in a series of small dots. The region containing each dot must then be individually excised and treated separately for incubations and washes.
Samples for dot blots
Protein samples for titrating antibodies should contain the protein of interest in abundance. Recombinant protein is ideal, however cell lysates containing highly expressed protein can also be used.
Negative control samples can also be included, particularly if cell lysates are used. Negative control samples will determine whether any observed signal is due to non-specific cross-reactivity.
Preparation of samples
Although sample preparation for dot blotting is similar to sample preparation for traditional Western blotting, several factors should be kept in mind. These apply both to when using microfiltration units or when spotting the protein manually.
- Prepare enough sample in sufficient volume to accommodate all the conditions being tested
- Do not prepare samples in buffers containing detergents as they will inhibit binding of the protein to the membrane
- If detergents are present, dilute the samples with buffer
- If a sample contains precipitates, centrifuge the sample and only apply the supernatant to membrane to prevent clogging
- Dilute viscous samples in buffer
Dot blot diagram/conditions
Titration of both primary and secondary antibodies can be performed simultaneously by using a checkerboard titration pattern. Figure 1 is an example of an experiment to titrate a primary antibody with a recommended dilution of 1:1000 and a secondary antibody with a recommended dilution of 1:10,000.
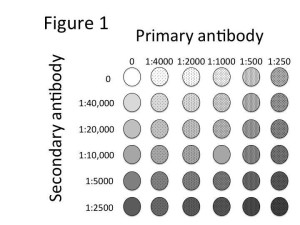
The following guidelines should be followed when planning the experiment:
- Create a diagram of experimental conditions (similar to Figure 1)
- Plan to titrate primary and secondary antibodies on opposing axes
- Choose ranges of antibody dilutions that encompass the recommended concentration and two dilutions above and below the recommended concentration. For example, for an antibody with a recommended dilution of 1:1000, use dilutions of 1:250, 1:500, 1:1000, 1:2000 and 1:4000
- Include spaces containing no primary or secondary antibodies to control for background
Dot blot procedure using a commercial apparatus
Commercial dot blot apparatuses immobilize, concentrate and bind samples to membranes using a vacuum to draw the sample onto the membrane. Washes and antibody incubations can also be performed using the units. Dot blot apparatuses can utilize either a slot blot template that binds the protein to the membrane in a thin slit or a round template for application of the protein in a circle. The slot format is preferred for densitometry and quantitation, however the round format is technically easier to work with because it decreases bubble formation.
Follow the manufacturer’s instructions to set up and prepare the apparatus.
General protocol
- Apply the sample in a volume large enough to cover the exposed membrane in each well
- Apply sample in the center of the well being careful to avoid creating air bubbles
- Do not exceed binding capacity of the membrane
- Close off unused wells by filling them with sample buffer
- Follow the manufacturer’s instructions to draw the sample through the membrane using the vacuum
- Perform washes and incubations according to the manufacturer’s guidelines
- Remove membrane from the unit and perform ECL detection using a standard kit
Dot blot procedure using a manual spotting method
A manual dot blot procedure follows the same principal as when using a dot blot apparatus, however the areas in which the proteins are spotted must be delineated by drawing a grid on the membrane. Due to sample diffusion, smaller volumes must be used when manually spotting proteins. After blotting, samples must be physically separated by cutting the membrane prior to incubation with antibodies.
- Delineate spotting areas by drawing a grid on the membrane using a pencil. Maintain a 1cm minimum distance between samples
- Build a stack to accommodate the membrane
- Place paper towels on work surface; enough towels should be used to keep bottom towels dry throughout procedure
- Place dry filter paper on top of paper towels
- Place filter paper prewet with buffer on dry filter paper
- Place prewet membrane on filter paper
- Spot samples onto membrane
- Spot 1-5 microliters of each sample onto center of each marked square in the grid marked on the membrane
- Choose a sample volume that will not spread between squares on the grid and will not exceed the binding capacity of the membrane
- For greater than 2 microliters, apply 2 microliters, allow sample to absorb, then add additional sample to same spot
- Do not exceed binding capacity of membrane
- Sample should wick into membrane and not spread across membrane. Membranes that are too wet will cause spreading of sample
- Place membrane on clean filter paper to dry after sample is absorbed
- Following the grid lines, cut a square around each circle and place each membrane into a separate container
- Fishing tackle boxes work well for titrating antibodies
- Wash and incubate each square with the specified primary and secondary antibody combinations following a standard Western blot protocol
- Perform ECL detection using a standard kit
Photo courtesy of Stinging Eyes.


